Protein is considered one of the three major macronutrients, the other two being fats and carbohydrates (with alcohol sometimes considered a fourth). Protein is typically consumed in moderate amounts on the ketogenic diet, but some experts recommend a higher protein diet. In order to understand the benefits of protein in your diet, you must first understand basic protein metabolism and physiological functions.
Protein Function
Dietary protein is essential to human life because it provides the substrate for the production of enzymes, hormones, antibodies, acid-base balance, energy, and structural components of the cell [1]. In addition, protein is required for the synthesis of new muscle tissue and provides the means for an increase in strength and hypertrophy.
Protein provides four kilocalories per gram and is structurally different from carbohydrates, fats, or alcohols because it contains a nitrogen group [2]. Carbohydrates, fats, and alcohols contain only hydrogen, oxygen, and carbon in different amounts depending on the specific nutrient and its function. Proteins, on the other hand, contain hydrogen, oxygen, carbon, and the addition of nitrogen. Amino acids are the individual subunits of protein; therefore, when protein is broken down via digestion, which I will talk about later, it is divided into each constituent amino acid.

Amino Acids- The Building Blocks of Protein
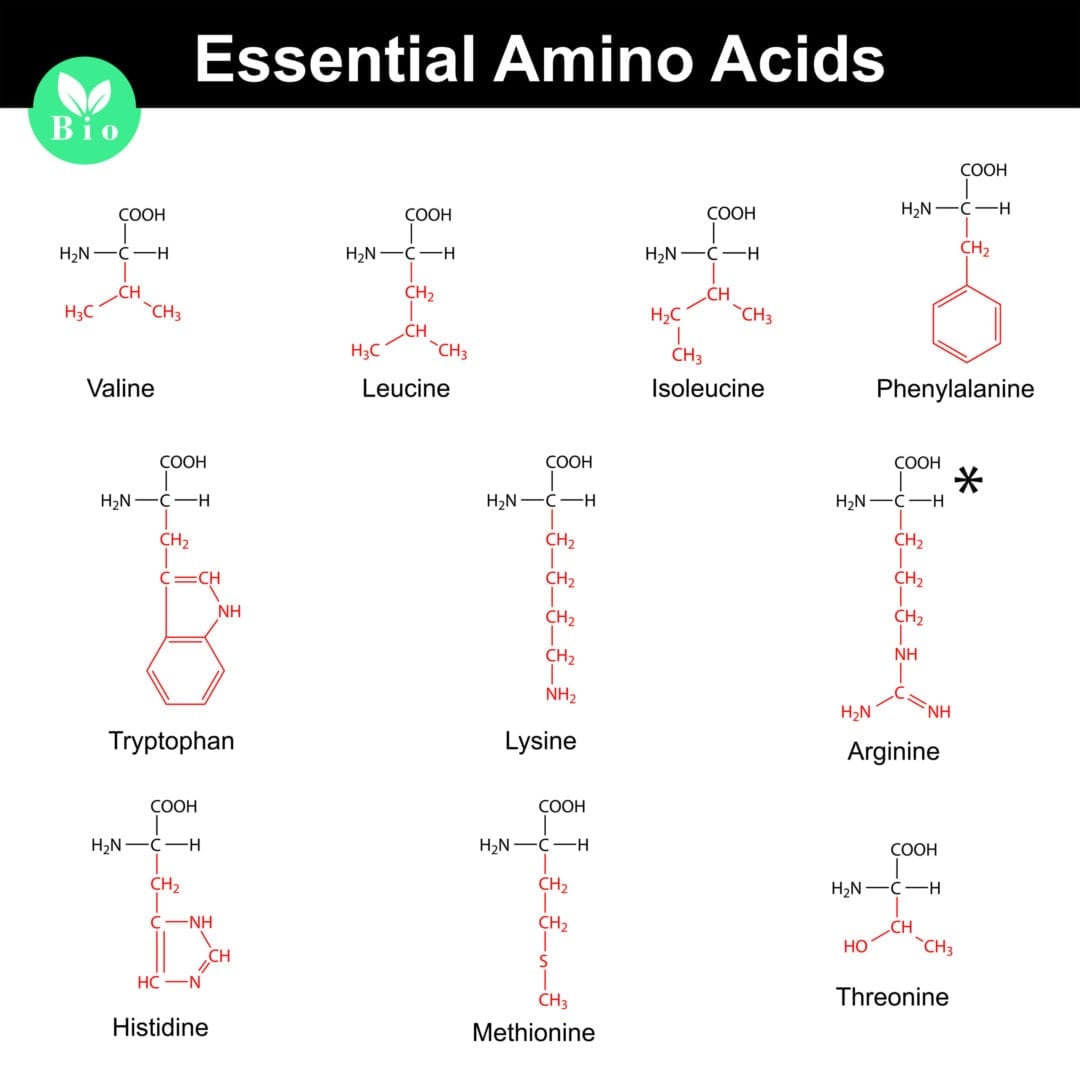
Among the pool of amino acids that make up protein, there are 20 that are further divided into two separate categories; essential and non-essential. Essential amino acids are those that your body cannot synthesize themselves; therefore, they must be consumed in the diet. Non-essential amino acids are those that can be synthesized by the body to carry out their necessary function. Non-essential amino acids can be synthesized in the body as long as there is enough substrate (essential amino acids) present [2].
Essential amino acids include Histidine, Lysine, Threonine, Isoleucine, Methionine, Tryptophan, Leucine, Phenylalanine, and Valine. Non-essential amino acids include Alanine, Aspartic Acid, Glutamine, Serine, Arginine, Cysteine, Glycine, Tyrosine, Asparagine, Glutamic Acid, and Proline.
Protein Metabolism
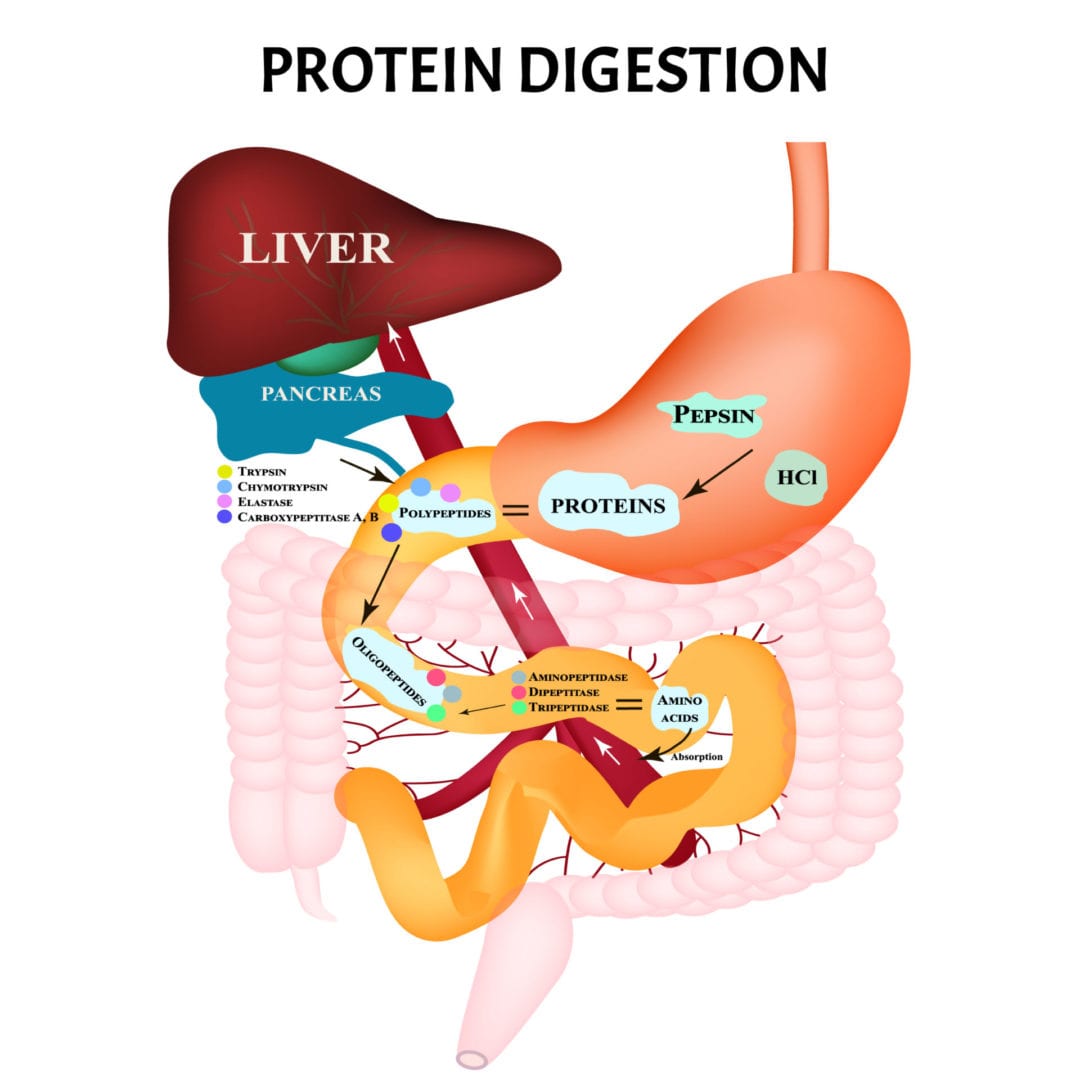
Upon ingestion, protein is mechanically digested (chewing) in the mouth. It then travels down the esophagus where it enters the stomach and is broken down by an enzyme called pepsin. The function of pepsin is to catalyze the breakdown of the peptide bonds that hold together the various amino acids making up the protein.
From here, the shorter chains of amino acids (polypeptides) are then transferred to the small intestine where the enzymes trypsin, chymotrypsin, and carboxypeptidase further begin to breakdown the peptide bonds holding the polypeptides together.
With assistance from brush border enzymes, which are enzymes found on the microvilli of the small intestine, these polypeptides are then broken down further into peptides or short chains of amino acids.
These amino acids are then absorbed into the bloodstream through the walls of the small intestine where they then travel to the liver to be used for their necessary function [3].
Enzyme, Hormone, and Antibody Production
Once at the liver, amino acids are sent to various parts of the body to begin the synthesis of other products necessary for proper bodily function. Amino acids can synthesize enzymes, which are what we call catalysts, meaning they cause a reaction in the body to occur. They are considered a tertiary protein structure and each is comprised of specific amino acids used to cause a reaction. In addition, amino acids can also form hormones, which are needed to initiate a process in a specific cell type. Antibodies, on the other hand, are comprised of polypeptides of amino acids (University of Arizona, 2000). These structures are made by white blood cells, or ß-cells, of the immune system and are important because they identify and destroy pathogens [4].
Acid-Base Balance
Another important function of amino acids in the maintenance of proper acid-base balance, or pH. In short, amino acids can be positively or negatively charged due to the hydrogen ions they contain. When the body becomes too acidic, for example in times of intense exercise, the protein will accept these excess hydrogen ions. Whereas when the body becomes too basic, these amino acids will donate their hydrogen ions to maintain equilibrium. The body’s pH is maintained around 7.35-7.45 on the pH scale. The pH scale ranges from 0 to 14; where 0 is highly acidic with a high concentration of hydrogen ions and 14 is highly basic due to the lack of hydrogen ions (University of Wisconsin). To reference, water is 7 or neutral, bleach is around 11 or highly basic, and vinegar is around 3 or highly acidic. The pH scale ranges from 0 being highly acidic with a high concentration of hydrogen ions to 14 being highly basic due to the lack of hydrogen ions (University of Wisconsin).

Energy
As mentioned previously, proteins do contain 4 kilocalories per gram, therefore, they yield energy. However, protein oxidation (the use of amino acids as fuel) is dependent on various conditions. In times of high stress and low stored energy, the body will oxidize protein to provide a substrate for more energy. This occurs largely when exercise has been maintained for an extended period of time without the consumption of either carbohydrates or fats.
Structural Components
Lastly, protein makes up some structural components of cells. The majority of the cell membrane is comprised of fatty acids called phospholipids. However, protein makes up the parts of the cell membrane that are responsible for the flow of nutrients across the cell membrane to be used within the cell [5].
Muscle Protein Synthesis
One of the most notable functions of protein is its ability to synthesis new muscle tissue. After the protein is broken down, it is sent to different parts of the body to perform a specific function. One of these functions is muscle protein synthesis (MPS). MPS is triggered by the amino acid, leucine, in addition to mechanical damage (lifting weights) or phospholipids (phosphatidic acid) [6]. When MPS this action is triggered, the body begins to take the ingested amino acids, codes them to be used to make new muscle, then translates these codes into new muscle tissue.
Concluding Thoughts on Protein Metabolism
As you can see protein metabolism is essential to many physiological functions within the body. These functions cannot occur without the presence of the necessary amino acids; therefore, it is crucial that protein is a staple in your diet.
Key Takeaways
- Protein is a macronutrient that provides 4 calories per gram.
- Protein can be broken down into amino acids.
- Amino acids can be classified as essential or non-essential.
- Protein is required for the production of enzymes, hormones, antibodies, acid-base balance, energy, and structural components of the cell.
- Protein can also provide energy as well as aid in the synthesis of new muscle.
References
Seebahor, Bob. (2011). Nutrition Periodization for Athlete’s: Taking Traditional Sports Nutrition to the Next Level. Boulder, Colorado. Bull Publishing Company
Mahan, L. K., Escott-Stump, S. (2012). Krause’s Food and Nutrition Therapy (12th Edition). St. Louis, Missouri. Saunders Elsevier.
Gillaspy, R. (2016). Protein Digestion and Absorption Process (Chapter 5, Lesson 14). Retrieved from http://study.com/academy/lesson/protein-digestion-and-absorption-process.html
Delves, P.J. (2016). Overview of the Immune System. Retrieved from https://www.merckmanuals.com/home/immune-disorders/biology-of-the-immune-system/overview-of-the-immune-system
Corsini, D. (2016). Major Structural Components of the Cell Membrane. Retrieved from http://study.com/academy/lesson/major-structural-components-of-the-cell-membrane.html
Joy, J.M., Lowery, R.P., Dudeck, J.E., De Souza, E.O., Ja?ger, R., McCleary, S.A., Wilson, S.M.C., Purpura, M., Wilson, J.M. (2013) Phosphatidic Acid Supplementation Increases Skeletal Muscle Hypertrophy and Strength. International Society of Sports Nutrition.