Post Traumatic Stress Disorder (PTSD)
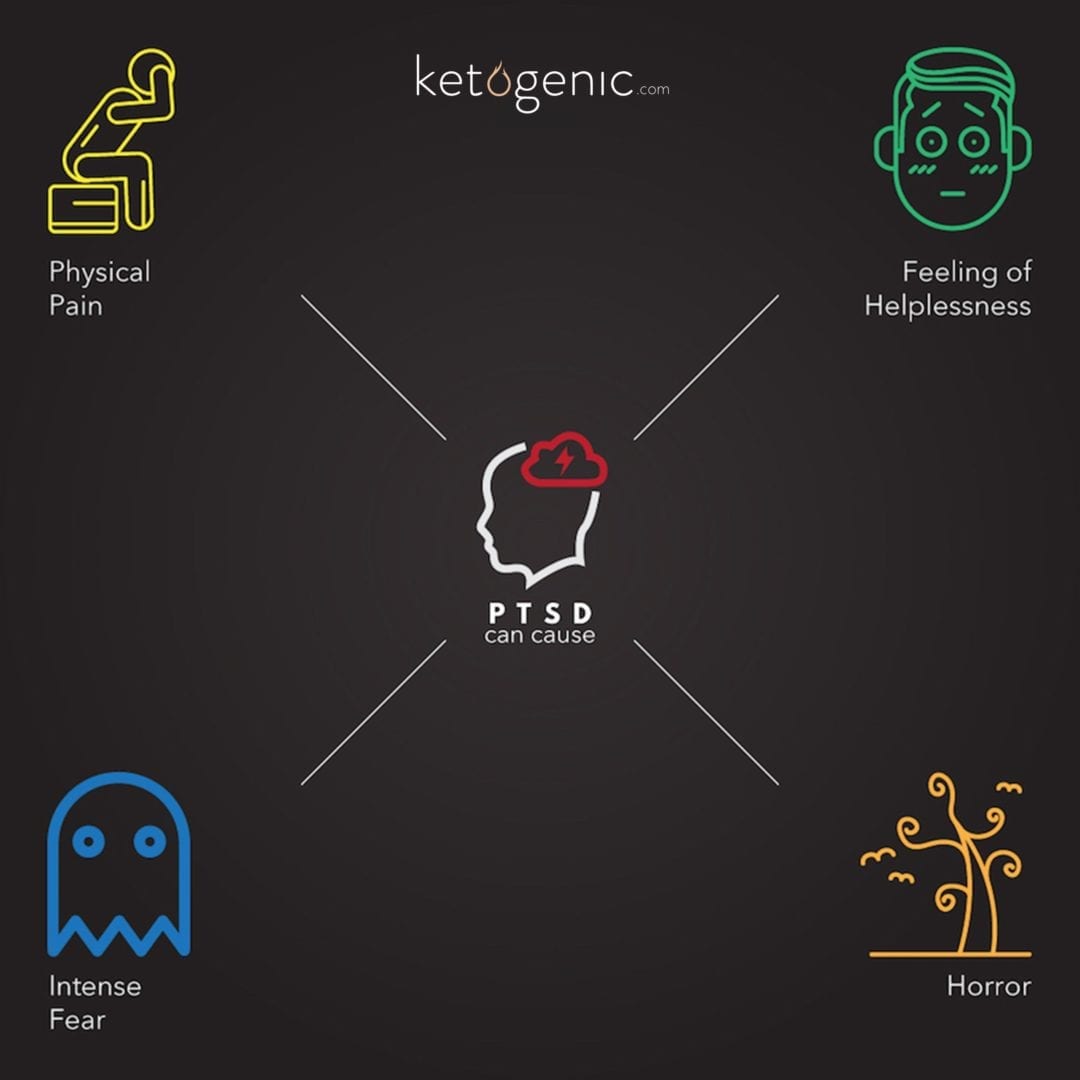 Post traumatic stress disorder (PTSD) is a mental disorder arising from a threatening event(s) that may cause physical harm, intense fear, feeling of helplessness, and/or horror. [1] PTSD is often accompanied by poor physical health and is prevalent in veterans as well as the general population. [2] [3] Although the data is limited, previous studies have supported the notion that PTSD-related impairments in physical health may result from a disrupted gut-brain axis. [4] [5] [6] [7]
Post traumatic stress disorder (PTSD) is a mental disorder arising from a threatening event(s) that may cause physical harm, intense fear, feeling of helplessness, and/or horror. [1] PTSD is often accompanied by poor physical health and is prevalent in veterans as well as the general population. [2] [3] Although the data is limited, previous studies have supported the notion that PTSD-related impairments in physical health may result from a disrupted gut-brain axis. [4] [5] [6] [7]
PTSD and the Gut-Brain Axis
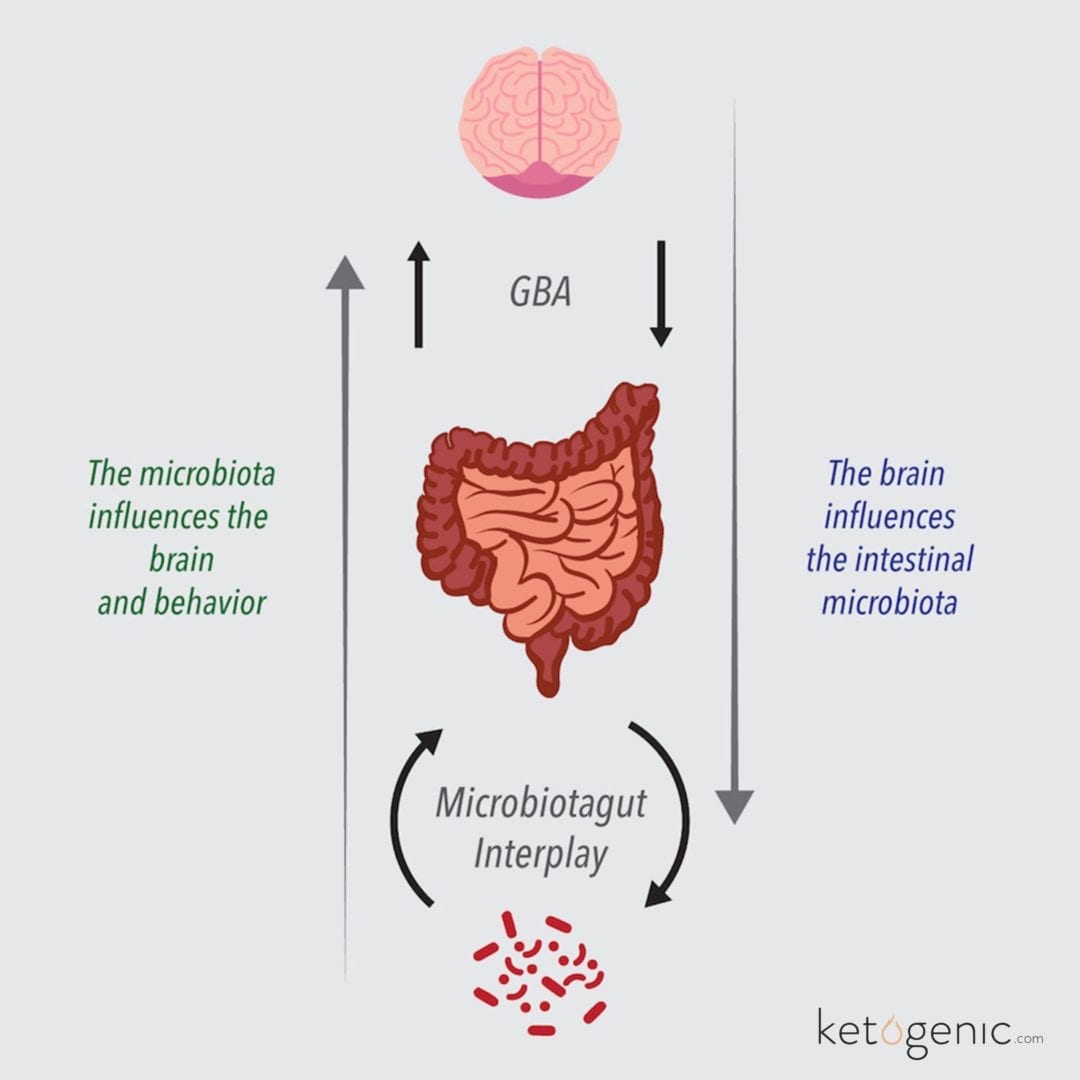
The brain and gastrointestinal system are extensively connected complex systems with similar pathways and neurotransmitters. [8] [9] Generally, a stressed brain leads to a dysfunctional gut, and a dysfunctional gut affects the brain – it is a vicious cycle. [8] [10] Mediators interlinking the brain and gut include the immune system, colon-dwelling microbiome, and circulating inflammation and hormones. [9] [10] In regards to the gut side of the gut-brain axis, individuals with PTSD commonly exhibit gastrointestinal dysfunction via irritable bowel syndrome, increased intestinal permeability, altered gut microbiota, altered stress hormone levels (i.e., cortisol), and increased systemic inflammation (i.e., C-reactive protein and lipopolysaccharides). [7] [11] [12] [13] [14] The brain side of the axis is also impaired with PTSD, demonstrated by signs of compromised cognitive performance (i.e., short-term memory and attention span) and emotional health (i.e., hostility levels, alcohol dependency, and domestic violence tendencies). [7] [15]
 Physiologic dysregulation that occurs with PTSD streams through the blood vessels of these diagnosed patients. Characteristically, PTSD is associated with elevated blood glucose levels as well as dyslipidemia defined as increases in triglyceride and low density lipoprotein cholesterol (LDL-c) levels with decreases in high density lipoprotein cholesterol (HDL-c) levels. [16] [17]The combination of dyslipidemia and elevated glucose levels places individuals with PTSD at an increased risk for developing type 2 diabetes. [18] [19] Furthermore, individuals diagnosed with PTSD demonstrate elevated basal heart rates and blood pressure, which contribute to the possible development of cardiovascular disease. [20] [21]
Physiologic dysregulation that occurs with PTSD streams through the blood vessels of these diagnosed patients. Characteristically, PTSD is associated with elevated blood glucose levels as well as dyslipidemia defined as increases in triglyceride and low density lipoprotein cholesterol (LDL-c) levels with decreases in high density lipoprotein cholesterol (HDL-c) levels. [16] [17]The combination of dyslipidemia and elevated glucose levels places individuals with PTSD at an increased risk for developing type 2 diabetes. [18] [19] Furthermore, individuals diagnosed with PTSD demonstrate elevated basal heart rates and blood pressure, which contribute to the possible development of cardiovascular disease. [20] [21]
PTSD Therapies and Treatment
Effective therapies in the treatment of PTSD are lacking. A plausible holistic therapy, due to its efficacy in improving PTSD-associated health deficiencies, is the incorporation of a ketogenic diet. The ketogenic diet is a high-fat, low-carbohydrate, moderate protein dietary regimen that has successfully treated numerous chronic diseases. Unlike a regular mixed diet which promotes only glucose as the primary fuel source for the brain, the ketogenic diet provides two primary energy sources for the brain: 1) glucose, derived from glycogen and gluconeogenesis in the liver, and 2) ketone bodies, synthesized from fatty acids in the liver. [22]
On a molecule-by-molecule basis, the ketone body beta-hydroxybutyrate (β-HB) provides more energy than glucose, and has been shown to be a beneficial fuel for the brain, especially in a damaged brain cell. [23] Neurological diseases and symptoms improved by a ketogenic diet include epilepsy, headaches, neurotrauma, Alzheimer’s disease, Parkinson’s disease, sleep disorders, brain cancer, autism, and multiple sclerosis. [24] [25] [26]
Keto and PTSD
 Cognitive performance and emotional health impairments often associated with PTSD may be improved by the ketogenic diet. The mechanisms and outcomes of a ketogenic diet are similar to fasting as both lack dietary glucose and subsequently produce ketone bodies to supply energy-requirements.ays) possibly due to increased β-HB levels and/or decreased glucose levels. [27] [28] β-HB is an isomer of γ-hydroxybutyrate (GHB), a euphoric drug that may be used as treatment for alcohol and opiate dependency and narcolepsy-associated cataplexy (sudden loss of control over muscles). [27] Similar to the actions of its isomer GHB, β-HB may also cause a mild euphoric state which subsequently improves mood and cognitive function. [27] Also, reduced cerebral glucose levels resulting from low carbohydrate consumption may promote neurogenesis and the synthesis of neurotrophic factors such as brain-derived neurotrophic factor (BDNF) that is associated with improved cognition, serotonin metabolism, and reduced aging of the brain. [29]
Cognitive performance and emotional health impairments often associated with PTSD may be improved by the ketogenic diet. The mechanisms and outcomes of a ketogenic diet are similar to fasting as both lack dietary glucose and subsequently produce ketone bodies to supply energy-requirements.ays) possibly due to increased β-HB levels and/or decreased glucose levels. [27] [28] β-HB is an isomer of γ-hydroxybutyrate (GHB), a euphoric drug that may be used as treatment for alcohol and opiate dependency and narcolepsy-associated cataplexy (sudden loss of control over muscles). [27] Similar to the actions of its isomer GHB, β-HB may also cause a mild euphoric state which subsequently improves mood and cognitive function. [27] Also, reduced cerebral glucose levels resulting from low carbohydrate consumption may promote neurogenesis and the synthesis of neurotrophic factors such as brain-derived neurotrophic factor (BDNF) that is associated with improved cognition, serotonin metabolism, and reduced aging of the brain. [29]
The repeated success that the ketogenic diet has shown with treating dyslipidemia and systemic inflammation makes it an excellent therapeutic candidate for PTSD. The ketogenic diet’s management of dyslipidemia occurs via a decrease in liver triglyceride synthesis (due to fewer carbohydrates consumed) [30] [31], a reduction of circulating triglyceride levels, and an elevation of HDL-c levels; these alterations decrease the risk for type 2 diabetes and cardiovascular disease. [32] [33] [34] [35] Additionally, the ketogenic diet has been associated with obesity prevention/reduction via reduced adiposity (specifically, reduced visceral adipose tissue) and reduced inflammation. [35] [36] [37] In addition to the brain, ketone bodies fuel other tissues including skeletal muscle, heart, and intestinal tissues. [38]Intestinal cells are called enterocytes; the luminal membranes of the large intestine enterocytes are laden with a layer of microorganisms that contribute to enhancing the immune system. Typically, this colon-dwelling microbiome consumes carbohydrates (i.e., fiber) and releases short chain fatty acids (SCFAs) as a by-product that subsequently fuels the enterocytes. [9] [10] Accordingly, with appropriate levels of fiber, the ketogenic diet may improve the health of the intestines by supplying enterocytes with two forms of energy: 1) SCFAs produced by the microbiome, and 2) ketone bodies synthesized by the liver. [9] [38]
In addition to the brain, ketone bodies fuel other tissues including skeletal muscle, heart, and intestinal tissues. [38]Intestinal cells are called enterocytes; the luminal membranes of the large intestine enterocytes are laden with a layer of microorganisms that contribute to enhancing the immune system. Typically, this colon-dwelling microbiome consumes carbohydrates (i.e., fiber) and releases short chain fatty acids (SCFAs) as a by-product that subsequently fuels the enterocytes. [9] [10] Accordingly, with appropriate levels of fiber, the ketogenic diet may improve the health of the intestines by supplying enterocytes with two forms of energy: 1) SCFAs produced by the microbiome, and 2) ketone bodies synthesized by the liver. [9] [38]
The compilation of the previous studies support the postulation that the ketogenic diet may ultimately improve gut, immune, and brain health to reestablish a well-functioning gut-brain axis. The efficacy of a ketogenic diet as a health intervention for those suffering from PTSD has not been studied in any major context. PTSD negatively affects many veterans as well as individuals of the general population, but the treatment for this diagnosis is lacking. Therefore, research studies that examine the effectiveness of dietary-based treatments for PTSD, such as the ketogenic diet, are of great importance.
References
Pitman, R. K., Rasmusson, A. M., Koenen, K. C., Shin, L. M., Orr, S. P., Gilbertson, M. W., … & Liberzon, I. (2012). Biological studies of post-traumatic stress disorder.Nature Reviews Neuroscience, 13(11), 769-787.
Friedman, M. J., & Schnurr, P. P. (1995). The relationship between trauma, post-traumatic stress disorder, and physical health.
Stam, R., Akkermans, L. M., & Wiegant, V. M. (1997). Trauma and the gut: interactions between stressful experience and intestinal function. Gut, 40(6), 704.
Solomon, Z., & Mikulincer, M. (1987). Combat stress reactions, post traumatic stress disorder and somatic complaints among Israeli soldiers.Journal of Psychosomatic Research, 31(1), 131-137.
Van der Ploeg, H. M., & Kleijn, W. C. (1989). Being held hostage in the Netherlands: A study of long-term aftereffects.Journal of Traumatic Stress,2(2), 153-169.
Fass, R., Kagan, B., Fullerton, S., & Mayer, E. A. (1995, April). The prevalence of functional gastrointestinal symptoms in male-pateints with PTSD. InGastroenterology (Vol. 108, No. 4, pp. A597-A597). Independence Square West Curtis Center, STE 300, Philadelphia, PA 19106-3399: WB Saunders Co.
Kharrazian, D. (2015). Traumatic Brain Injury and the Effect on the Brain-Gut Axis.Altern Ther Health Med, 28-32.
Romijn, J. A., Corssmit, E. P., Havekes, L. M., & Pijl, H. (2008). Gut–brain axis.Current Opinion in Clinical Nutrition & Metabolic Care, 11(4), 518-521.
Bercik, P., Collins, S. M., & Verdu, E. F. (2012). Microbes and the gut‐brain axis.Neurogastroenterology & Motility, 24(5), 405-413.
Cryan, J. F., & O’Mahony, S. M. (2011). The microbiome‐gut‐brain axis: from bowel to behavior.Neurogastroenterology & Motility, 23(3), 187-192.
Söderholm, J. D., & Perdue, M. H. (2001). II. Stress and intestinal barrier function.American Journal of Physiology-Gastrointestinal and Liver Physiology, 280(1), G7-G13.
Yehuda, R. (2002). Post-traumatic stress disorder.New England Journal of Medicine, 346(2), 108-114.
Rohleder, N., Joksimovic, L., Wolf, J. M., & Kirschbaum, C. (2004). Hypocortisolism and increased glucocorticoid sensitivity of pro-Inflammatory cytokine production in Bosnian war refugees with posttraumatic stress disorder.Biological psychiatry, 55(7), 745-751.
Leclercq, S., Forsythe, P., & Bienenstock, J. (2016). Posttraumatic Stress Disorder Does the Gut Microbiome Hold the Key?.The Canadian Journal of Psychiatry, 0706743716635535.
American Psychiatric Association. (2003). APA (2000).Diagnostic and statistical manual of mental disorders, 4.
Karlović, D., Buljan, D., Martinac, M., & Marčinko, D. (2004). Serum lipid concentrations in Croatian veterans with post-traumatic stress disorder, post-traumatic stress disorder comorbid with major depressive disorder, or major depressive disorder.Journal of Korean medical science, 19(3), 431-436.
Cohen, B. E., Marmar, C., Ren, L., Bertenthal, D., & Seal, K. H. (2009). Association of cardiovascular risk factors with mental health diagnoses in Iraq and Afghanistan war veterans using VA health care.JAMA, 302(5), 489-492.
Norman, S. B., Means‐Christensen, A. J., Craske, M. G., Sherbourne, C. D., Roy‐Byrne, P. P., & Stein, M. B. (2006). Associations between psychological trauma and physical illness in primary care.Journal of traumatic stress,19(4), 461-470.
Flood, A. M., Boyle, S. H., Calhoun, P. S., Dennis, M. F., Barefoot, J. C., Moore, S. D., & Beckham, J. C. (2010). Prospective study of externalizing and internalizing subtypes of posttraumatic stress disorder and their relationship to mortality among Vietnam veterans.Comprehensive psychiatry, 51(3), 236-242.
Buckley, T. C., Holohan, D., Greif, J. L., Bedard, M., & Suvak, M. (2004). Twenty-four-hour ambulatory assessment of heart rate and blood pressure in chronic PTSD and non-PTSD veterans.Journal of Traumatic Stress, 17(2), 163-171.
Coughlin, S. S. (2011). Post-traumatic stress disorder and cardiovascular disease.The open cardiovascular medicine journal, 5(1).
Cahill Jr, G. F., & Owen, O. E. (1968). Starvation and survival.Transactions of the American Clinical and Climatological Association, 79, 13.
Cox, P. J., & Clarke, K. (2014). Acute nutritional ketosis: implications for exercise performance and metabolism.Extreme physiology & medicine,3(1),
Hartman, A. L., Gasior, M., Vining, E. P., & Rogawski, M. A. (2007). The neuropharmacology of the ketogenic diet.Pediatric neurology, 36(5), 281-292.
McDaniel, S. S., Rensing, N. R., Thio, L. L., Yamada, K. A., & Wong, M. (2011).The ketogenic diet inhibits the mammalian target of rapamycin (mTOR) pathway.Epilepsia, 52(3), e7-e11.
Stafstrom, C. E., & Rho, J. M. (2012). The ketogenic diet as a treatment paradigm for diverse neurological disorders.Front Pharmacol, 3(59), 1-5.
Brown, A. J. (2007). Low-carb diets, fasting and euphoria: Is there a link between ketosis and γ hydroxybutyrate (GHB)?.Medical hypotheses, 68(2), 268-271.
Fond, G., Macgregor, A., Leboyer, M., & Michalsen, A. (2013). Fasting in mood disorders: neurobiology and effectiveness. A review of the literature. Psychiatry research, 209(3), 253-258.
Fontán-Lozano, Á., López-Lluch, G., Delgado-García, J. M., Navas, P., & Carrión, Á. M. (2008). Molecular bases of caloric restriction regulation of neuronal synaptic plasticity.Molecular neurobiology, 38(2), 167-177.
Hellerstein, M. K. (1999). De novo lipogenesis in humans: metabolic and regulatory aspects.European journal of clinical nutrition, 53, S53-S65.
Paoli, A., Rubini, A., Volek, J. S., & Grimaldi, K. A. (2013). Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets.European journal of clinical nutrition, 67(8), 789-796.
Sharman, M. J., Kraemer, W. J., Love, D. M., Avery, N. G., Gómez, A. L., Scheett, T. P., & Volek, J. S. (2002). A ketogenic diet favorably affects serum biomarkers for cardiovascular disease in normal-weight men.The Journal of nutrition, 132(7), 1879-1885.
Volek, J. S., Sharman, M. J., & Forsythe, C. E. (2005). Modification of lipoproteins by very low-carbohydrate diets.The Journal of nutrition, 135(6), 1339-1342.
Paoli, A., Canato, M., Toniolo, L., Bargossi, A. M., Neri, M., Mediati, M., … & Bianco, A. (2010). The ketogenic diet: an underappreciated therapeutic option. La Clinica Terapeutica, 162(5), e145-53.
Holland, A. M., Kephart, W. C., Mumford, P. W., Mobley, C. B., Lowery, R. P., Shake, J. J., …& Huggins, K. W. (2016). Effects of a ketogenic diet on adipose tissue, liver and serum biomarkers in sedentary rats and rats that exercised via resisted voluntary wheel running.American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, ajpregu-00156.
Kahn, S. E., Hull, R. L., & Utzschneider, K. M. (2006). Mechanisms linking obesity to insulin resistance and type 2 diabetes.Nature, 444(7121), 840-846.
Van Gaal, L. F., Mertens, I. L., & Christophe, E. (2006). Mechanisms linking obesity with cardiovascular disease.Nature, 444(7121), 875-880.
Robinson, A. M., & Williamson, D. H. (1980). Physiological roles of ketone bodies as substrates and signals in mammalian tissues.Physiological reviews, 60(1), 143-187.